1. Introduction
A complex interrelated function is present between kidneys and the thyroid gland which has significant clinical implications. A major role in metabolizing, degrading, and excreting thyroid hormones is played by the kidney, while the thyroid hormones are of importance for renal embryogenesis, growth, and normal renal physiological functioning [1].
Chronic kidney disease (CKD) is global public health issue of increasing concern in the past decade, that approximately affects 10–15 % of the population worldwide. The National Kidney Foundation’s Kidney Disease Outcome Quality Initiative (K/DOQI) defines CKD as the renal abnormalities that persists for more than three months with or without decreased glomerular filtration rate (GFR), progressing through stages resulting in end-stage kidney disease (ESKD) [1]. Hemodialysis is the modulator of ESKD management worldwide, which sustains millions of patients [11].
Several studies concluded that patients with advanced CKD mainly those on maintenance hemodialysis, are more likely to have thyroid abnormalities in comparison to age-matched population with normal renal function [12].
Biochemical abnormalities are not the only complication of thyroid dysfunction in CKD patients; it was stated that these patients have an increased cardiovascular morbidity, renal failure progression, as well as increased risk of mortality [13–15]. Confounding factors including altered protein-binding, hemodialysis effects, and metabolites accumulation limit thyroid functions in CKD [9,16].
2. Methodology
2.1 Search strategy
According to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines, a systematic literature review was conducted. PubMed/MEDLINE, Scopus, Web of science, and Google Scholar were the electronic database used to search for studies published between January 1, 2020, and March 15, 2026.
2.2 Inclusion and Exclusion criteria
Inclusion Criteria:
- Study published between January 2020 and March 2026
- Study examines thyroid function in adult patients (≥ 18 years) with CKD (any stage) or undergoing hemodialysis
- Studies reporting prevalence, determinants, clinical outcomes, or management of thyroid dysfunction
- Published in English
- Full-text availability
Exclusion Criteria:
- Published before 2020
- Case reports, case series (<10 patients), editorials, commentaries, and conference abstracts
- Studies focused exclusively on pediatric populations
- Duplicate publications or overlapping cohorts
3. Results
3.1 Study Selection and Characteristics
The primary result of the database search was 847 potentially relevant record. 613 articles underwent title and abstract screening after removing the duplicates (n=234). Full-text review of 97 articles yielded and resulted in 34 studies meeting the mentioned inclusion criteria. Their characteristics are summarized in Table 1
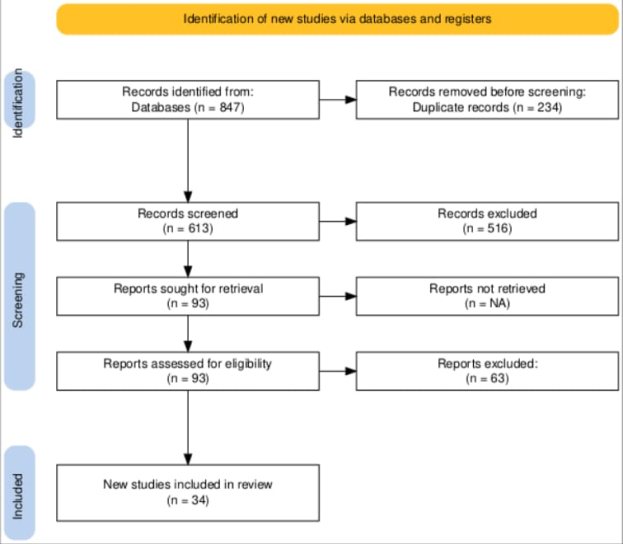
Fig. 1. PRISMA Flow Diagram
Table 1
Characteristics of Included studies
|
First Author |
Year |
Country |
Study Design |
Population |
Sample Size |
Key Focus |
|
Adani |
2023 |
Somalia |
Cross-sectional |
Hemodialysis |
301 |
Prevalence, determinants |
|
Agahi |
2024 |
Iran |
Review |
CKD all stages |
N/A |
Thyroid-renal interrelationship |
|
Ansari |
2023 |
India |
Cross-sectional |
CKD stages 1–5 |
200 |
Thyroid dysfunction by stage |
|
Asmar |
2023 |
Lebanon |
Cohort (10-year) |
Hemodialysis |
188 |
Anemia, mortality |
|
Burton |
2021 |
UK |
Review |
Hemodialysis |
N/A |
Advances in hemodialysis |
|
Cheng |
2025 |
China |
Cross-sectional |
CKD stages 1–5 |
1,842 |
CKD-thyroid hormones |
|
Farhana |
n.d |
India |
Cross-sectional |
CKD |
100 |
Thyroid profile |
|
German |
2025 |
Pakistan |
Cross-sectional |
ESKD |
185 |
Thyroid dysfunction |
|
Gomba |
2024 |
Nigeria |
Cross-sectional |
CKD |
200 |
Prevalence, patterns |
|
Hafed |
2024 |
Saudi Arabia |
Cross-sectional |
CKD stages 3–5 |
164 |
Thyroid function |
|
Haja |
2025 |
India |
Cross-sectional |
CKD |
120 |
Thyroid hormone status |
|
Huang |
2020 |
USA |
Cohort |
CKD (age ≥55) |
195,417 |
Hypothyroidism-CKD association |
|
Inaba |
2021 |
Japan |
Cross-sectional |
Hemodialysis |
686 |
FT3/FT4 ratio |
|
Kashif |
2023 |
India |
Cross-sectional |
CKD |
200 |
Thyroid dysfunction |
|
Kim |
2023 |
Korea |
Population-based |
General with CKD |
3,765 |
Subclinical thyroid dysfunction |
|
Matsuoka-Uchiyama |
2022 |
Japan |
Cross-sectional |
CKD |
422 |
Hypothyroidism-proteinuria |
|
Narasaki |
2021 |
USA |
Review |
CKD |
N/A |
Thyroid-kidney interplay |
|
Obasuyi |
2023 |
Nigeria |
Cross-sectional |
CKD |
160 |
Spectrum of dysfunction |
|
Otsuka |
2024 |
Japan |
Retrospective cohort |
Hemodialysis |
98 |
Roxadustat and hypothyroidism |
|
Pujitha Mallina |
2025 |
India |
Cross-sectional |
CKD |
200 |
Renal function with/without hypothyroidism |
|
Raj |
2023 |
India |
Cross-sectional |
CKD |
150 |
Thyroid abnormalities |
|
Ratiu |
2026 |
Romania |
Cross-sectional |
Hemodialysis |
112 |
Drivers of hypothyroidism |
|
Sanai |
2021 |
Japan |
Prospective |
Hemodialysis |
50 |
Hemodilution effects |
|
Scandelai |
2025 |
Brazil |
Cross-sectional |
CKD stages 3–4 |
86 |
Thyroid function-proteinuria |
|
Schultheiss |
2021 |
Germany |
Cohort (GCKD study) |
CKD stages 2–4 |
5,045 |
Thyroid function, renal events |
|
Shakya |
2023 |
India |
Cross-sectional |
ESKD |
108 |
Thyroid-ESKD interactions |
|
Sinjari |
2022 |
Iraq |
Cross-sectional |
CKD |
120 |
Thyroid function disorders |
|
Spahia |
2023 |
Kosovo |
Review |
CKD |
N/A |
Subclinical hypothyroidism |
|
Topal |
2023 |
Turkey |
Cross-sectional |
Hemodialysis |
98 |
Klotho, anemia |
|
Xu, W |
2021 |
China |
Cross-sectional |
CKD stages 3–5 |
280 |
Thyroid autoantibodies, CVD |
|
Xu, Y |
2024 |
USA |
Cross-sectional + MR |
General population |
12,548 |
Hypothyroidism-CKD relationship |
|
You |
2022 |
USA |
Prospective cohort |
Hemodialysis |
145 |
TSH, endothelial dysfunction |
|
Yuasa |
2020 |
Japan |
Cross-sectional |
CKD stages 1–5 |
448 |
Hypothyroidism prevalence |
|
Yuasa |
2023 |
Japan |
Cross-sectional |
CKD |
60 |
Urinary thyroid hormone excretion |
|
Zhang |
2022 |
China |
Retrospective |
CKD with crescents |
168 |
FT3/FT4 ratio as biomarker |
|
Zhao |
2025 |
China |
Review |
Dialysis/CKD |
N/A |
Thyroid hormone replacement |
|
Zheng |
2023 |
China |
Cohort |
Renal anemia |
210 |
Roxadustat, thyroid function |
3.2 Prevalence of Thyroid Dysfunction in CKD
3.2.1 Overall Estimate of Prevalence
|
Study |
Population |
Country |
Hypothyroidism (%) |
Subclinical Hypothyroidism (%) |
Overt Hypothyroidism (%) |
Low T3 Syndrome (%) |
|
Hemodialysis/ESKD Populations | ||||||
|
Adani 2023 |
Hemodialysis |
Somalia |
28 |
16.0 (57.8 % of hypothyroid) |
11.6 (42.2 % of hypothyroid) |
NR |
|
German 2025 |
ESKD |
Pakistan |
31.4 |
24.3 |
7 |
NR |
|
Inaba 2021 |
Hemodialysis |
Japan |
NR |
NR |
NR |
Reduced FT3/FT4 ratio common |
|
Shakya 2023 |
ESKD |
India |
33.3 |
23.1 |
10.2 |
41.70 % |
|
Ratiu 2026 |
Hemodialysis |
Romania |
25.9 |
19.6 |
6.3 |
33.00 % |
|
You 2022 |
Hemodialysis |
USA |
Elevated TSH common |
NR |
NR |
NR |
|
CKD All Stages | ||||||
|
Ansari 2023 |
CKD 1–5 |
India |
32 |
22 |
10 |
15.50 % |
|
Cheng 2025 |
CKD 1–5 |
China |
23.1 |
17.3 |
5.8 |
28.40 % |
|
Gomba 2024 |
CKD |
Nigeria |
26.5 |
20.5 |
6 |
18.00 % |
|
Haja 2025 |
CKD |
India |
29.2 |
21.7 |
7.5 |
NR |
|
Kashif 2023 |
CKD |
India |
30.5 |
23 |
7.5 |
NR |
|
Obasuyi 2023 |
CKD |
Nigeria |
27.5 |
20 |
7.5 |
31.30 % |
|
Raj 2023 |
CKD |
India |
28.7 |
20.7 |
8 |
22.00 % |
|
Yuasa 2020 |
CKD 1–5 |
Japan |
19.2 |
14.5 |
4.7 |
NR |
|
Early CKD (Stages 1–3) | ||||||
|
Huang 2020 |
CKD (age≥55) |
USA |
12.8 |
NR |
NR |
NR |
|
Kim 2023 |
CKD |
Korea |
14.2 |
11.8 |
2.4 |
NR |
|
Scandelai 2025 |
CKD 3–4 |
Brazil |
20.9 |
17.4 |
3.5 |
NR |
|
Schultheiss 2021 |
CKD 2–4 |
Germany |
10.5 |
8.7 |
1.8 |
NR |
|
Xu, Y. 2024 |
General with CKD |
USA |
11.3 |
NR |
NR |
NR |
Between 25.9–33.3 % of the cases of hypothyroidism in patients undergoing hemodialysis were subclinical hypothyroidism, making it the majority (57.8–77.4 %). According to Adani et al. [1] hypothyroidism was found in 28 % of Somali hemodialysis patients, of which 57.8 % of them were exhibiting subclinical and 42.2 % over hypothyroidism. German et al. [17] showed 31.4 % hypothyroidism among Pakistani ESKD patients (24.3 % subclinical, 7.0 % overt), while in Shakya et al. [9] 33.3 % in Indian ESKD patients (23.1 %, 10.2 % overt)
Non-CKD populations had a prevalence of hypothyroidism ranging between 10.5–32.0 %, which shows a clear association with the disease severity [13, 18]. Ansari et al. [19] documented 32.0 % overall hypothyroidism across CKD stages 1–5 in India. Low T3 syndrome (euthyroid sick syndrome) was found out to be highly prevalent, affecting 15.5–41.7 % of CKD patients [5, 20, 21].
3.2.2 Prevalence by CKD Stage
A consistent pattern was found across the studies: thyroid dysfunction prevalence shows an abrupt increase and progression with the renal function decline [19,22]. Adani et al. [1] cited the Third National Health and Nutrition Examination Survey (NHANES), stating hypothyroidism prevalence of 5.4 %, 10.9 %, 20.4 %, 23.0 %, and 23.1 % among individuals with eGFRs of ≥90, 60–89, 45–59, 30–44, and <30 mL/min/1.73m², respectively.
According to Yuasa et al. [7] Japanese patients had a prevalence of hypothyroidism in CKD stage 3–12.3 %, CKD stage 4–18.7 %, and 24.5 % in stage 5. In a Korean study carried by Kim et al. [12] found an increase of prevalence with declining eGFR. Matsuoka-Uchiyama [23] and Scandelai et al. [16] have also confirmed the reduced kidney function and higher rates of hypothyroidism.
4. Discussion
This comprehensive review synthesizes evidence from 34 studies published between 2020–2026, providing updated insights into the complex relationship between thyroid dysfunction and chronic kidney disease [8,24]. The findings confirm that thyroid abnormalities, particularly hypothyroidism, are highly prevalent across the CKD spectrum and correlate with disease severity.
4.1 Prevalence in Context
Estimates of prevalence synthesized in the CKD population with thyroid dysfunction (25.9–33.3 % in hemodialysis, 10.5–32.0 % in CKD) surpasses those in the general population (4–10 % hypothyroidism), which is a confirmation that CKD is of high-risk significance for thyroid dysfunction [12,25]. Subclinical hypothyroidism predominance [26] does suggest that thyroid failure traditional mechanisms may be supplemented as well by uremia-related functional disturbances.
4.2 Clinical Significance of Determinants
Modifiable risks factors, such as, hypoalbuminemia, inflammation, and proteinuria, when identified can be potential intervention targets [16,27]. Theoretically nutritional support aimed to improve these factors can improve thyroid function, though prospective validation is required. Thyroid dysfunction was found to be associated with anemia, cardiovascular events and mortality [13,18].
Lower creatinine and overt hypothyroidism association [1,28] challenges the conventional understanding and highlights the unique physiology of hemodialysis patients. This can be possibly explained by the reduce in muscle mass (sarcopenia), which associated with hypothyroidism and is common in dialysis, or by the differential clearance of creatinine in hypothyroid versus euthyroid states. Another novel proposed mechanism contributing to hypothyroidism in proteinuric CKD patients is the loss of thyroid hormones in urine [10].
Roxadustat-induced central hypothyroidism emergence [6,7] shows the great importance in considering iatrogenic, as this medication is gaining wider use. Thyroid function monitoring must be done vigilantly by clinicians in patients receiving Roxadustat, along with differentiating the effect of drug from primary thyroid disease. The role of thyroid autoantibodies and biomarkers such as FT3/FT4 ratio has been explored by some studies to predict outcomes [15, 29].
5. Conclusion
This comprehensive review demonstrates the high prevalence of thyroid dysfunction, particularly hypothyroidism, in the CKD patients, affecting 10–15 % approximately in early CKD patients, and 25–33 % in those with end-stage disease [2, 3]. Subclinical hypothyroidism being predominant, by accounting for 60–75 % of the cases [1, 4, 30].
The clinical implications are substantial, with thyroid dysfunction linked to cardiovascular disease, endothelial dysfunction, anemia, nutritional abnormalities, and increased mortality [14,19,31]. Regular screening is warranted, with careful attention to the confounding effects of uremia, medications, and dialysis procedures on thyroid function test interpretation [9, 16].
Future research should focus on prospective interventional trials to determine the benefits of treating subclinical hypothyroidism, development of CKD-specific diagnostic criteria, and elucidation of optimal management strategies for special situations such as roxadustat-associated dysfunction [5, 10, 32].
References:
- Prevalence and Determinants of Hypothyroidism in Patients on Routine Hemodialysis in Somalia: A Cross-Sectional Study / A. A. Adani, M. O. Siyad, A. M. Adan, M O O Jeele. — Текст: непосредственный // Int J Gen Med. — 2023. — № 16. — С. 905–13.
- Chronic Kidney Disease and Thyroid Hormones / Y. Cheng, H. Hu, W. Li [и др.]. — Текст: непосредственный // J Clin Endocrinol Metab. — 2025. — № 110(8). — С. e2446–55.
- Elevated serum thyrotropin levels and endothelial dysfunction in a prospective hemodialysis cohort / A. S. You, M. Budoff, I. Zeb [и др.]. — Текст: непосредственный // Hemodial Int. — 2022. — № 26(1). — С. 57–65.
- Assessment of Thyroid Hormone Status Among Patients With Chronic Kidney Disease in a Tertiary Care Center: A Cross-Sectional Study / A. Haja, B. M. Rizwan, J. Michael [и др.]. — Текст: непосредственный // Cureus. — 2025. — № 17(12).
- Sinjari, H. Y. Thyroid Function Disorders in Patients with Chronic Kidney Disease / H. Y. Sinjari, J. M. Ibrahim. — Текст: непосредственный // Medical Journal of Babylon. — 2022. — № 19(1). — С. 76–80.
- Understanding the Drivers of Hypothyroidism in Patients Undergoing Chronic Hemodialysis / I. A. Ratiu, E. E. Babeș, L. M. Georgescu [и др.]. — Текст: непосредственный // Diagnostics (Basel). — 2026. — № 16(2):177.
- Prevalence of hypothyroidism in Japanese chronic kidney disease patients / R. Yuasa, Y. Ohashi, A. Saito [и др.]. — Текст: непосредственный // Ren Fail. — 2020. — № 42(1). — С. 572–9.
- Assessment of Renal Function in Patients with Chronic Kidney Disease with and without Hypothyroidism / Mallina Pujitha, Rajan Vinay, Kumar Eswar, Prasad Gullipalli. — Текст: непосредственный // Ethiop J Health Sci. — 2025. — № 35(1).
- Evaluation of Interactions Between Thyroid Dysfunction in End-Stage Renal Disease Patients: A Cross-Sectional Study / S. Shakya, S. Kumar, V. Verma [и др.]. — Текст: непосредственный // Cureus. — 2023.
- Urinary excretion of thyroid hormone in CKD patients: a proof-of-concept of nephrogenic hypothyroidism / R. Yuasa, M. Muramatsu, A. Saito [и др.]. — Текст: непосредственный // Ren Fail. — 2023. — № 45(2).
- Recent advances in treatment of haemodialysis / J. O. Burton, R. W. Corbett, P. A. Kalra [и др.]. — Текст: непосредственный // J R Soc Med. — 2021. — № 114(1). — С. 30–7.
- Subclinical thyroid dysfunction and chronic kidney disease: a nationwide population-based study / H. J. Kim, S. J. Park, H. K. Park [и др.]. — Текст: непосредственный // BMC Nephrol. — 2023. — № 24(1). — С. 64.
- Thyroid function, renal events and mortality in chronic kidney disease patients: the German Chronic Kidney Disease study / U. T. Schultheiss, I. Steinbrenner, M. Nauck [и др.]. — Текст: непосредственный // Clin Kidney J. — 2021. — № 14(3). — С. 959–68.
- Relationship between hypothyroidism and chronic kidney disease: Results from the National Health and Nutrition Examination Survey 2007 to 2012 and Mendelian randomization study / Y. Xu, X. Wang, G. Wang [и др.]. — Текст: непосредственный // Medicine. — 2024. — № 51. — С. e40925.
- Topal, M. The association of soluble Klotho levels with anemia and hemoglobin variability in hemodialysis patients / M. Topal, I. Guney. — Текст: непосредственный // Semin Dial. — 2023. — № 36(2). — С. 142–6.
- Correlation between thyroid function and proteinuria in patients with chronic kidney disease stages 3 and 4 / K. S. Scandelai, P D B F A Vianna, J P B Sanches [и др.]. — Текст: непосредственный // Int Urol Nephrol. — 2025. — № 12. — С. 4309–14.
- Thyroid Dysfunction in Patients With End-Stage Renal Disease: A Single-Centered Experience From Pakistan / S. German, S. Bhatti, T. Waqar [и др.]. — Текст: непосредственный // Cureus. — 2025. — № 1.
- Correlation between thyroid autoantibodies and cardiovascular disease in patients with stages 3–5 chronic kidney disease / W. Xu, S. Liang, Y. Huang [и др.]. — Текст: непосредственный // Ann Transl Med. — 2021. — № 9(16). — С. 1301.
- Thyroid Dysfunction at Different Stages of Chronic Kidney Disease: A Cross-Sectional Study at a Rural Teaching College in Central India / I. Ansari, S. Kumar, S. Acharya [и др.]. — Текст: непосредственный // Cureus. — 2023. — № 7. — С. e42130.
- Hemodilution Impacts Assessment of Thyroid Status before and after Hemodialysis in Patients with End-Stage Renal Disease / T. Sanai, K. Okamura, T. Onoue [и др.]. — Текст: непосредственный // Am J Nephrol. — 2021. — № 51(12). — С. 988–94.
- The Prevalence of Thyroid Abnormalities in Patients With Chronic Kidney Disease: A Cross-Sectional Study at a Tertiary Care Hospital / R. Raj, V. Kumar, D. Bhushan [и др.]. — Текст: непосредственный // Cureus. — 2023. — № 8.
- Thyroid Dysfunction and Chronic Kidney Disease: A Study Among the Northeastern Population of India / M. Kashif, M. S. Hussain, M. Anis, P. K. Shah. — Текст: непосредственный // Cureus. — 2023. — № 15(5).
- The association between hypothyroidism and proteinuria in patients with chronic kidney disease: a cross-sectional study / N. Matsuoka-Uchiyama, K. Tsuji, Y. Sang [и др.]. — Текст: непосредственный // Sci Rep. — 2022. — № 12(1). — С. 14999.
- Interrelationship between thyroid hormones and reduced renal function, a review article / S. Agahi, A. Amouzegar, M. Honarvar [и др.]. — Текст: непосредственный // Thyroid Res. — 2024. — № 17(1).
- Association between hypothyroidism and chronic kidney disease observed among an adult population 55 years and older / C. W. Huang, B. H. Li, K. Reynolds [и др.]. — Текст: непосредственный // Medicine. — 2020. — № 17. — С. e19569.
- Subclinical Hypothyroidism, Kidney, and Heart from Normal to Uremic Milieu / N. Spahia, M. Rroji, M. Barbullushi, G. Spasovski. — Текст: непосредственный // Metab Syndr Relat Disord. — 2023. — № 21(8). — С. 415–25.
- Obasuyi, J. O. Spectrum of Thyroid Dysfunction in Patients with Chronic Kidney Disease in Benin City, Nigeria / J. O. Obasuyi, M. A. Emokpae. — Текст: непосредственный // Medicines. — 2023. — № 10(8). — С. 47.
- Association of Reduced Free T3 to Free T4 Ratio with Lower Serum Creatinine in Japanese Hemodialysis Patients / M. Inaba, K. Mori, Y. Tsujimoto [и др.]. — Текст: непосредственный // Nutrients. — 2021. — № 13(12).
- The serum free triiodothyronine to free thyroxine ratio as a potential prognostic biomarker of chronic kidney disease in patients with glomerular crescents: A retrospective study / L. Zhang, Y. Wu, Y. Nie [и др.]. — Текст: непосредственный // Front Endocrinol (Lausanne). — 2022. — № 13.
- Assessment of Thyroid Function in Chronic Kidney Disease Patients at King Abdulaziz Medical City / A. B. Hafed, R. K. Abdulkareem, A. M. Almalki [и др.]. — Текст: непосредственный // Cureus. — 2024. — № 11.
- Roxadustat has risks of reversible central hypothyroidism in patients undergoing hemodialysis: a single-center retrospective cohort study / E. Otsuka, M. Kitamura, S. Funakoshi [и др.]. — Текст: непосредственный // Ren Fail. — 2024. — № 46(2). — С. 2410375.
- Gomba, V. E. Prevalence and patterns of thyroid dyfunction in chronic kidney disease patients in a tertiary hospital in southern Nigeria / V. E. Gomba, C. Unachukwu, S. Chinenye. — Текст: непосредственный // Int J Res Med Sci. — 2024. — № 12(3). — С. 651–7.

